As humanity prepares to return to deep space with missions like Artemis II, understanding how the human body responds to spaceflight is becoming essential. These missions are not only advancing exploration but also transforming how we study human biology beyond Earth.
Recent coverage of Artemis II has also highlighted how missions like this could help transform the future of space medicine, bringing human biology research into deep space in entirely new ways.
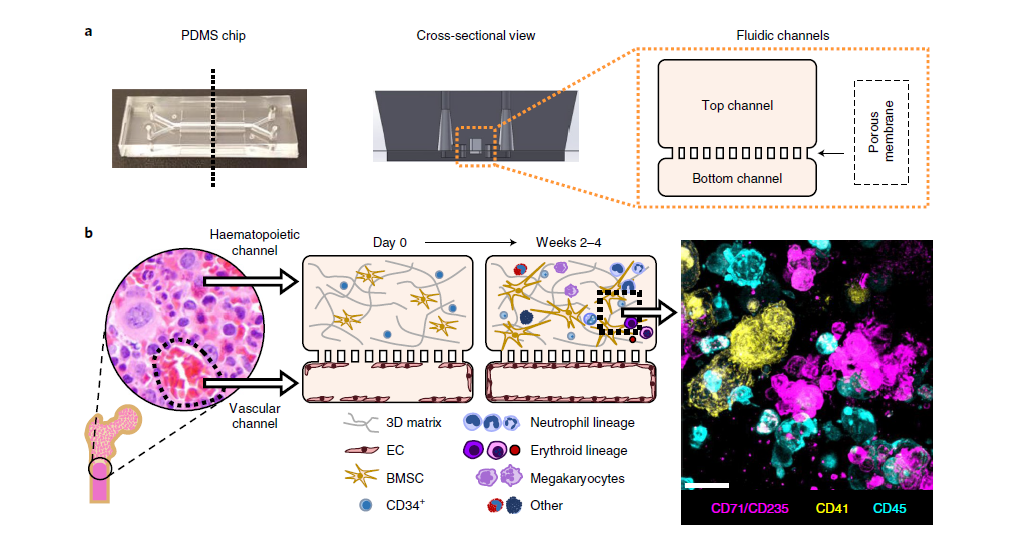
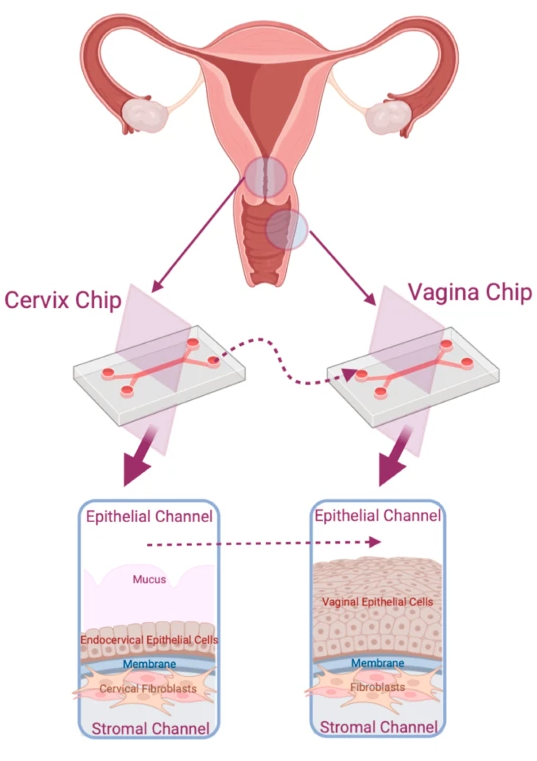
The AVATAR (A Virtual Astronaut Tissue Analog Response) program is a collaboration between NASA, the Wyss Institute, Emulate, Space Tango, and partners across the industry and US government. By leveraging advanced Organ-on-a-Chip technology, AVATAR aims to answer one of the most critical and complex questions in space biology: how does spaceflight affect human bone marrow?
Read below as we unpack this question and more with Ela Contreras-Panta, PhD, one of the key scientists working cross-functionally at Emulate and the Wyss Institute for Biologically Inspired Engineering at Harvard University, to bring the AVATAR program to space.
—
Hello Ela, thank you so much for taking the time to chat with us during this extremely busy time for you and the team. Let’s dive in with an easy one:
How does it feel to be a part of this historic moment?
Ela Contreras-Panta: The AVATAR project has been incredibly exciting for our team and colleagues. Working with these systems on the ground and knowing that parallel models are traveling beyond Earth adds a unique sense of connection between the lab and spaceflight that is hard to put into words.
It is monumental to be a part of an effort that brings together space exploration and human biology in a way that feels like sci-fi but is very much real life!
Why study bone marrow in space?
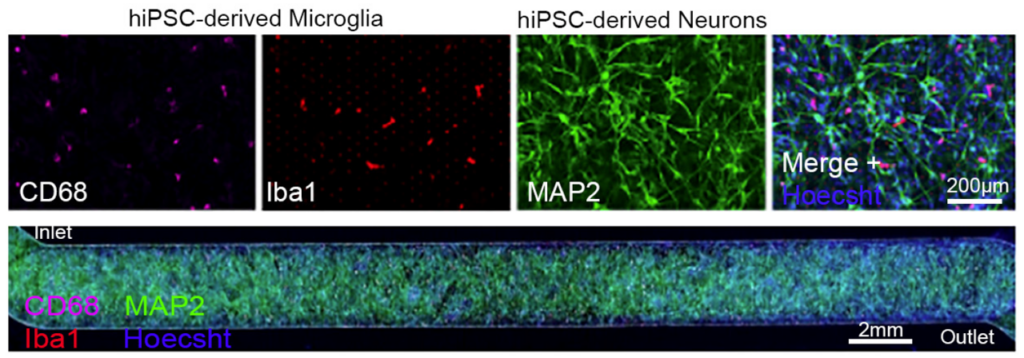
ECP: Bone marrow is the engine of the human hematopoietic system, responsible for producing the blood and immune cells that sustain life. It is also one of the most sensitive tissues to space-related stressors: radiation and microgravity.
Beyond Earth’s protective magnetic field, astronauts are exposed to higher levels of cosmic radiation, alongside the physiological challenges of microgravity. These conditions can disrupt immune function, alter blood cell production, and increase long-term health risks.
Despite its importance, bone marrow has historically been difficult to study in space, as traditional models cannot fully capture the complexity of human hematopoiesis. This creates a critical gap in our understanding, one that AVATAR is designed to address.
Why use the Organ-on-a-Chip approach?
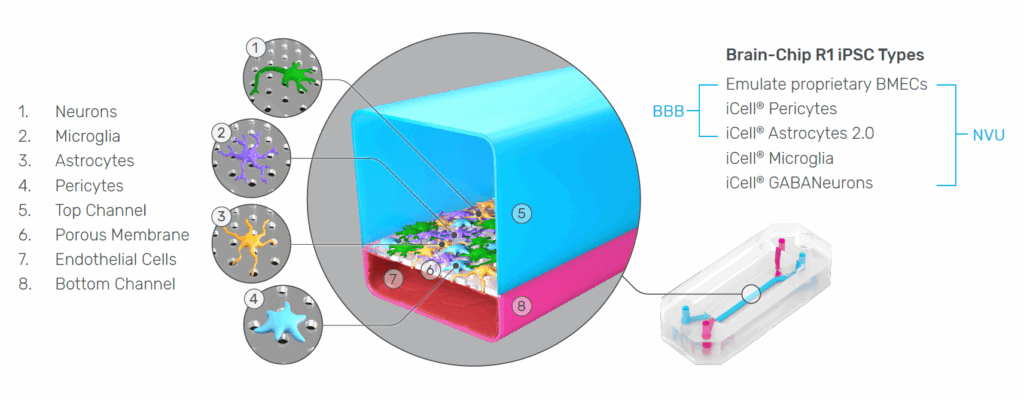
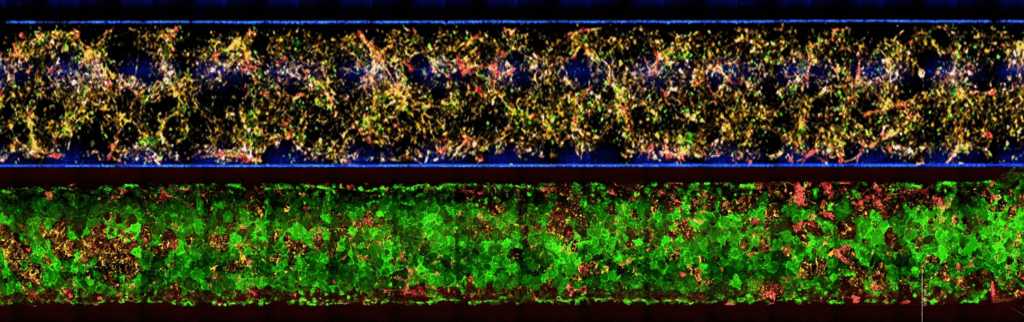
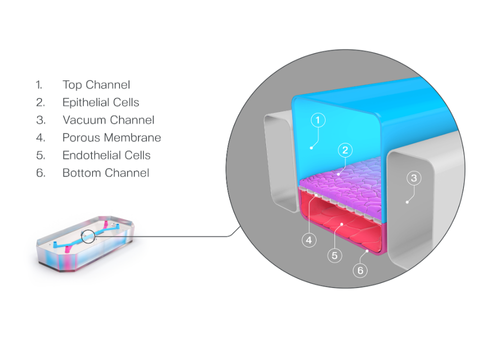
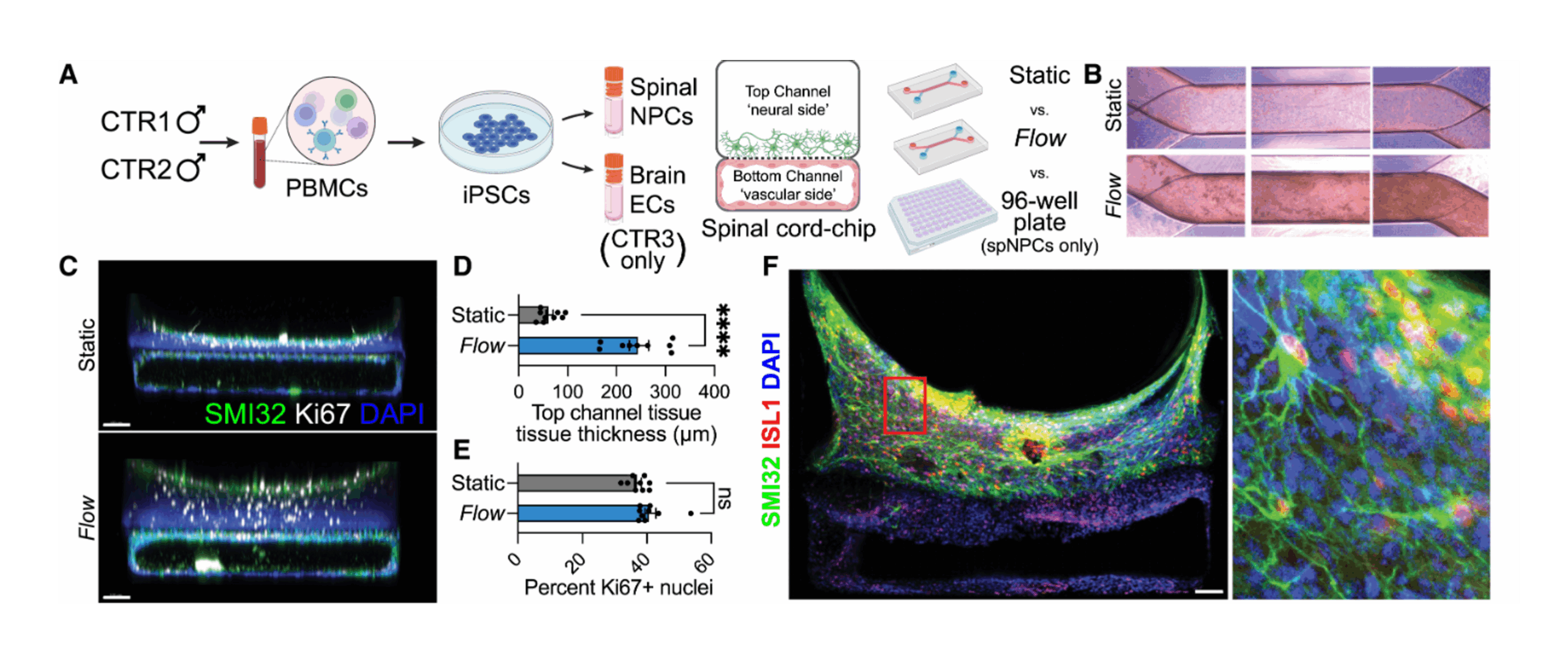
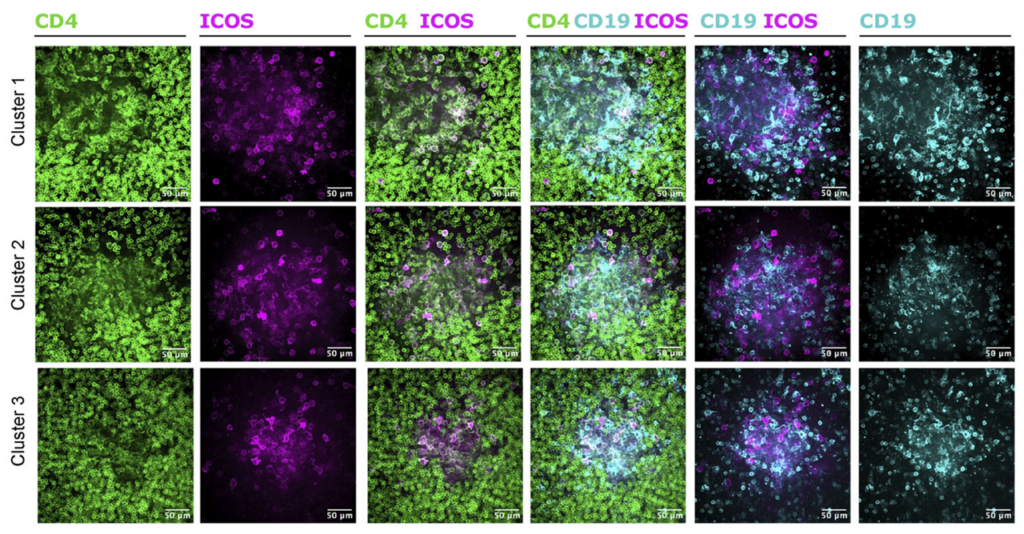
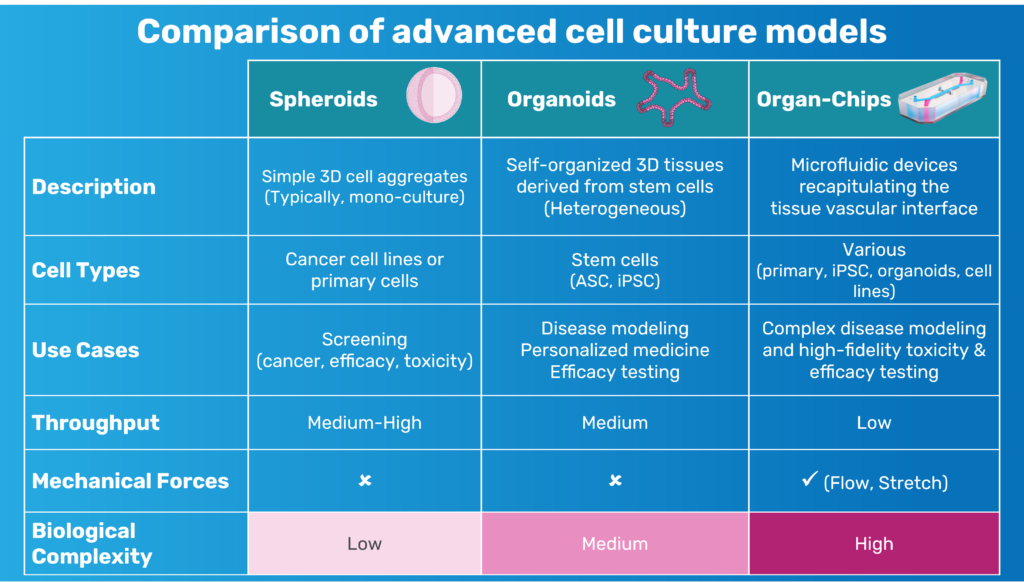
ECP: Organ-on-a-Chip technology offers a powerful solution to this challenge. These microfluidic systems recreate the architecture and function of living human tissues in vitro, allowing researchers to study dynamic biological processes in a controlled environment.
In AVATAR, these chips go one step further: they are built using cells derived directly from the Artemis II astronauts themselves. Each chip acts as a personalized “miniature AVATAR,” designed to mimic how that specific individual’s bone marrow may respond to spaceflight.
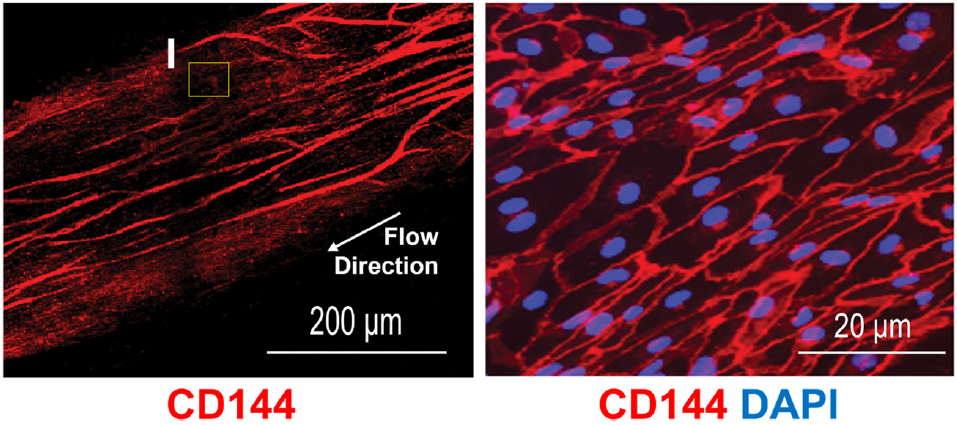
Emulate’s and Wyss Institute’s bone marrow Organ-Chip replicates key features of the human bone marrow niche, including cellular composition and continuous perfusion. By sending these chips into space alongside their donors, researchers can directly compare how human biology responds in deep space versus on Earth, which is something that has never been achieved before.
What more can you share about the experiment that’s in lunar orbit?
ECP: The AVATAR investigation flew aboard NASA’s Artemis II mission, a ~10-day journey around the moon that took astronauts beyond low-Earth orbit for the first time in over 50 years.
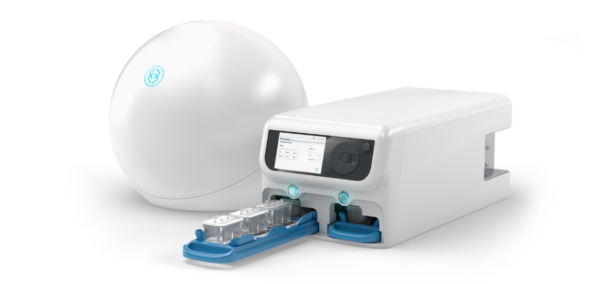
Inside the Orion spacecraft, the Organ-Chips are housed within Space Tango’s CubeLab™ platform. The CubeLab™ is an automated microgravity research system that maintains tightly controlled physiological conditions, including temperature and fluid flow, enabling the bone marrow chips to function as living tissue models throughout the mission.
After returning to Earth, the samples that were exposed to the effects of microgravity and deep space radiation will be analyzed at the molecular level to understand how thousands of genes and cellular pathways responded to space conditions. These results will be compared with parallel ground controls and astronaut blood samples, enabling a uniquely comprehensive view of human adaptation to space.
How could this research impact human health?
ECP: Beyond its immediate applications in astronaut health, AVATAR represents a shift in how we study human biology under extreme conditions.
By combining Organ-on-a-Chip technology with the unique environment of deep space, the project creates opportunities to uncover biological mechanisms that are difficult, or even impossible, to observe on Earth. These insights could accelerate the development of new therapeutic strategies, improve our ability to model complex diseases, and refine how we evaluate drug responses in human-relevant systems.
One of the most transformative, and one of the most exciting from my perspective, aspects of AVATAR is its focus on precision health. Because each Organ-Chip is derived from an individual astronaut, scientists can study how different people respond to the same space environment. This enables researchers to better understand individual variability and develop more targeted countermeasures for future missions.
AVATAR not only helps prepare us for future space exploration but also contributes to a broader transformation in biomedical research and personalized medicine.
How will this shape the future of space biology?
ECP: As we plan for the future, we hope that the AVATAR project paves the way for more Organ-on-a-Chip technology experiments in space.
As Organ-on-a-Chip technologies continue to evolve, future missions may incorporate multiple organ systems, larger cohorts of personalized chips, and even real-time data collection from deep space. Researchers may one day send AVATARS ahead of astronauts to predict health risks before a mission even begins, transforming how we prepare for human exploration beyond Earth.
This shift—from reacting to health challenges in space to proactively understanding and preventing them—marks a new chapter in both space medicine and biomedical research.
More broadly, the intersection of space exploration and biotechnology is unlocking entirely new possibilities for science. Through the AVATAR investigation, we are bringing human-relevant models into deep space, helping us better understand how life adapts beyond Earth, while also advancing research that can benefit human health here on the ground.
Being part of this effort has been incredibly meaningful, and I am so excited to see how the insights we gain will shape the future of both space exploration and medicine.
Thank you, Ela!
—
To learn more about the AVATAR investigation and its role in Artemis II, we invite you to explore NASA’s official resources and follow along as this journey unfolds.