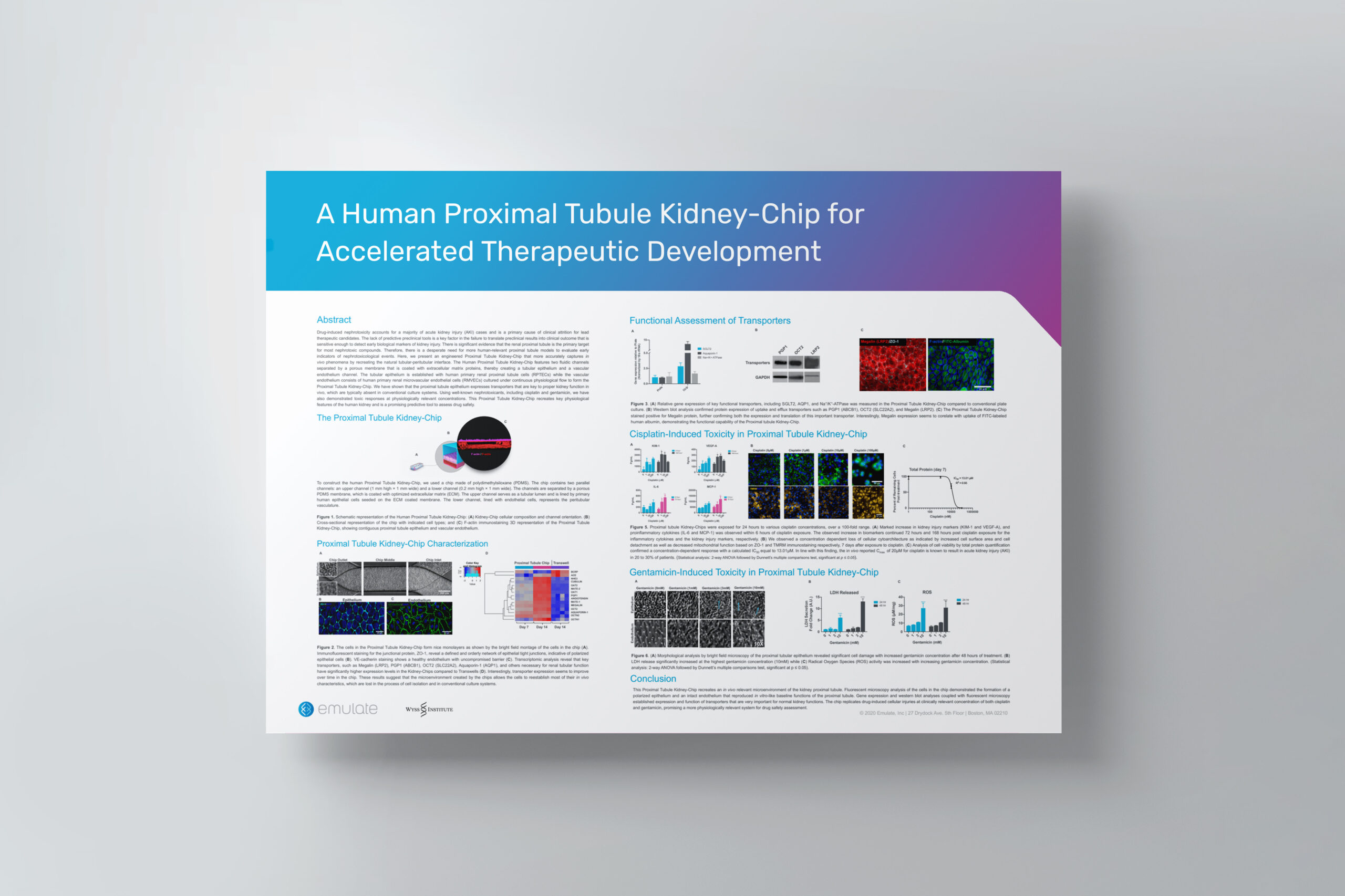
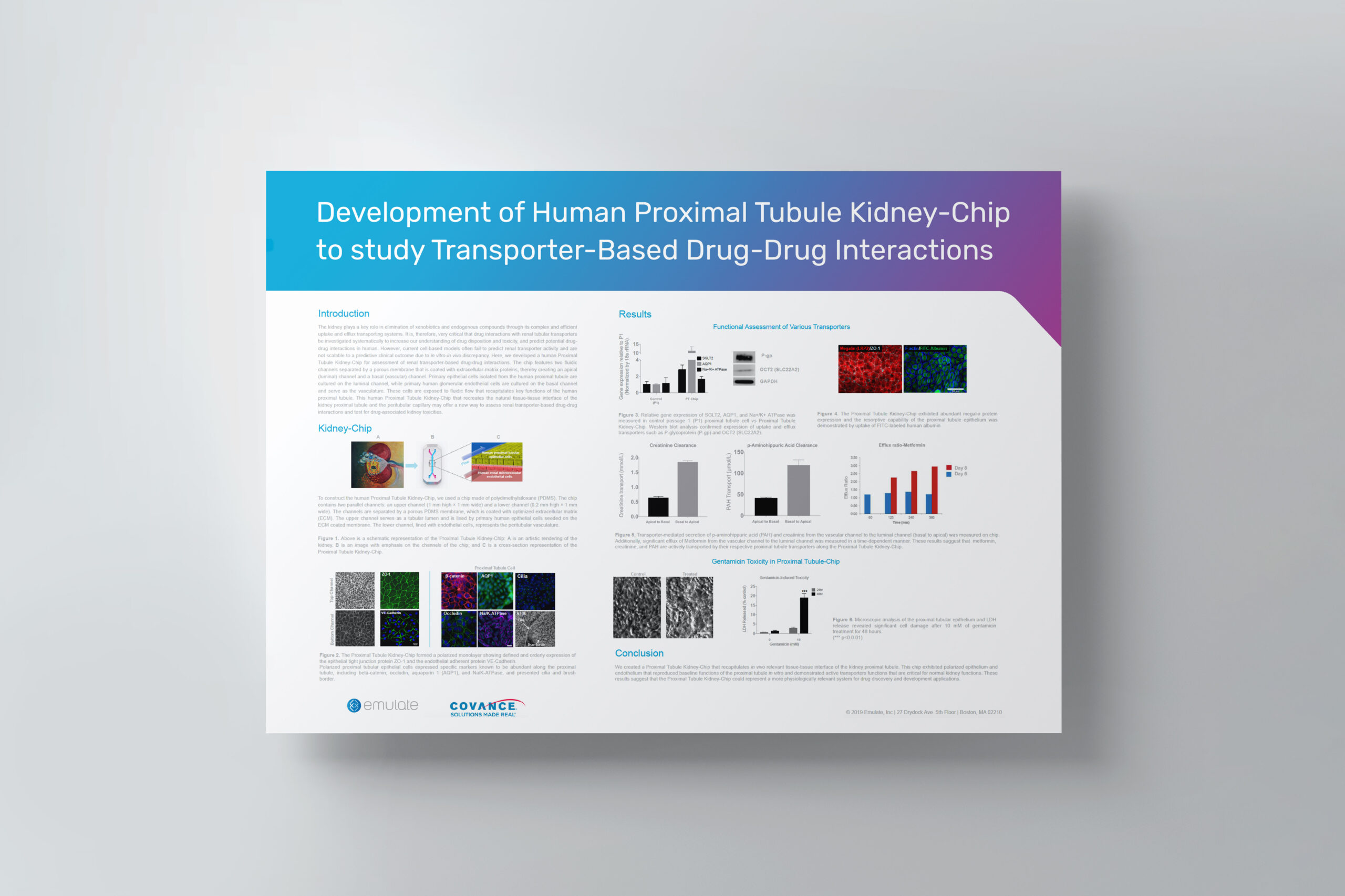
organ-Chip model
Kidney-Chip
Evaluate drug candidate toxicity at clinically relevant concentrations in a co-culture human kidney model


The Kidney-Chip is a BioKit Model that Emulate has internally developed and validated. It is available as a BioKit, which includes pre-qualified cells, Organ-Chip consumables, and validated protocols, with guarantees on characterization and functionality.
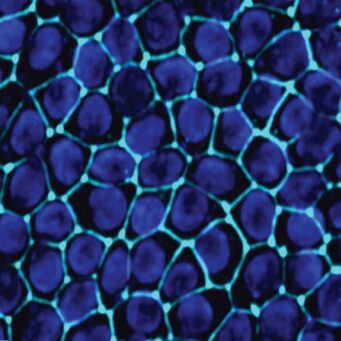
Characterization
A physiologically relevant kidney model
Inside the Kidney-Chip, cells achieve an in vivo-like phenotype, with high differentiation, normal epithelial cell polarity and morphology, and demonstrated functional transporter activity. This allows for a more physiological analysis of healthy kidney function and the nephrotoxicity of drug candidates. Long-term culture enables users to take multiple measurements for mechanistic studies, biomarker discovery, and nutrient metabolism.
Benefits
Contains key renal cell populations
The Kidney-Chip includes primary human proximal tubule epithelial cells and renal microvascular endothelial cells, enabling cell-cell interactions unlike monoculture cell models.
Retains important renal characteristics
The Kidney-Chip maintains functionality—including albumin reabsorption and characteristic cell morphology—for up to 14 days in culture, unlike conventional cell lines which lose differentiation over time.
Improved cytoarchitecture and polarization
Shear stress from media flow significantly improves epithelial cytoarchitecture, with greater polarization, cell height, and cilia formation than kidney epithelial cells in static culture.
Enhanced transporter activity
Sodium/phosphate (Na/Pi) co-transporter expression is increased on the Kidney-Chip in the presence of kidney-specific endothelial cells—effects not seen in mono-culture models or co-cultures with non-kidney-specific endothelial cells.
Application: Toxicology
Predicting drug-induced nephrotoxicity remains a challenge in preclinical development
Established preclinical in vitro models have limited utility in predicting early indicators of nephrotoxicity due to the inability to recreate the dynamic in vivo microenvironment. Cell lines have diminished transporter expression and functionality over time and are often not sensitive enough to respond to clinically relevant drug concentrations. Meanwhile, animal models have species differences in renal transporters, drug pharmacokinetics and pharmacodynamics, and metabolic responses. Taken together, improved preclinical kidney models are needed to improve patient safety.

Benefits
An improved preclinical model of nephrotoxicity for better clinical translation
Unlike conventional in vitro models, the Kidney-Chip can model mechanisms of drug-induced nephrotoxicity at clinically relevant drug concentrations. Side-by-side studies demonstrate the Kidney-Chip has better concentration-dependent responses than static epithelial monoculture models. A diverse array of endpoints can be measured, including morphological damage, cell death (LDH, ALP), oxidative stress, and a kidney injury panel (KIM-1, clusterin, TFF3, VEGF).
.
Compatible with Zoë-CM2® Culture Module
The Zoë-CM2 Culture Module is a versatile system for model development & target validation. Capable of culturing up to 12 Organ-Chips at a time, the user-friendly platform gives researchers a window into the inner workings of human biology.

Get Started Today
Experience the predictive power of Organ-on-a-Chip technology. Gain deeper insights by incorporating the Kidney-Chip in your lab workflow by using our BioKit or using your own cell sources with a Basic Research Kit.
FEATURED RESOURCE
Proximal Tubule Kidney-Chip for Modeling Human Physiology
Learn how the Proximal Tubule Kidney-Chip can be applied to emulate the complex functionality of the human kidney.