RECREATING DRUG SIDE-EFFECTS USING ORGANS-ON-CHIPS
Maintenance of the fluidity of blood within the circulatory system is an important physiological process that is tightly regulated by the endothelial cells that line the blood vessels of the human body. The term thrombosis usually refers to pathological blood clotting that can manifest clinically in relation to dysfunction of the circulatory system. As more therapeutics have been developed, it has also become apparent that some drugs can contribute to the development of thrombosis, resulting in serious adverse events.
It is difficult, however, to predict drug-induced thrombosis using current pre-clinical cell culture and animal models, due to the dynamic nature of the flow of blood in the body and the complex molecular mechanisms that regulate drug interaction with different blood components of the human circulatory system.

Human-Relevant Models
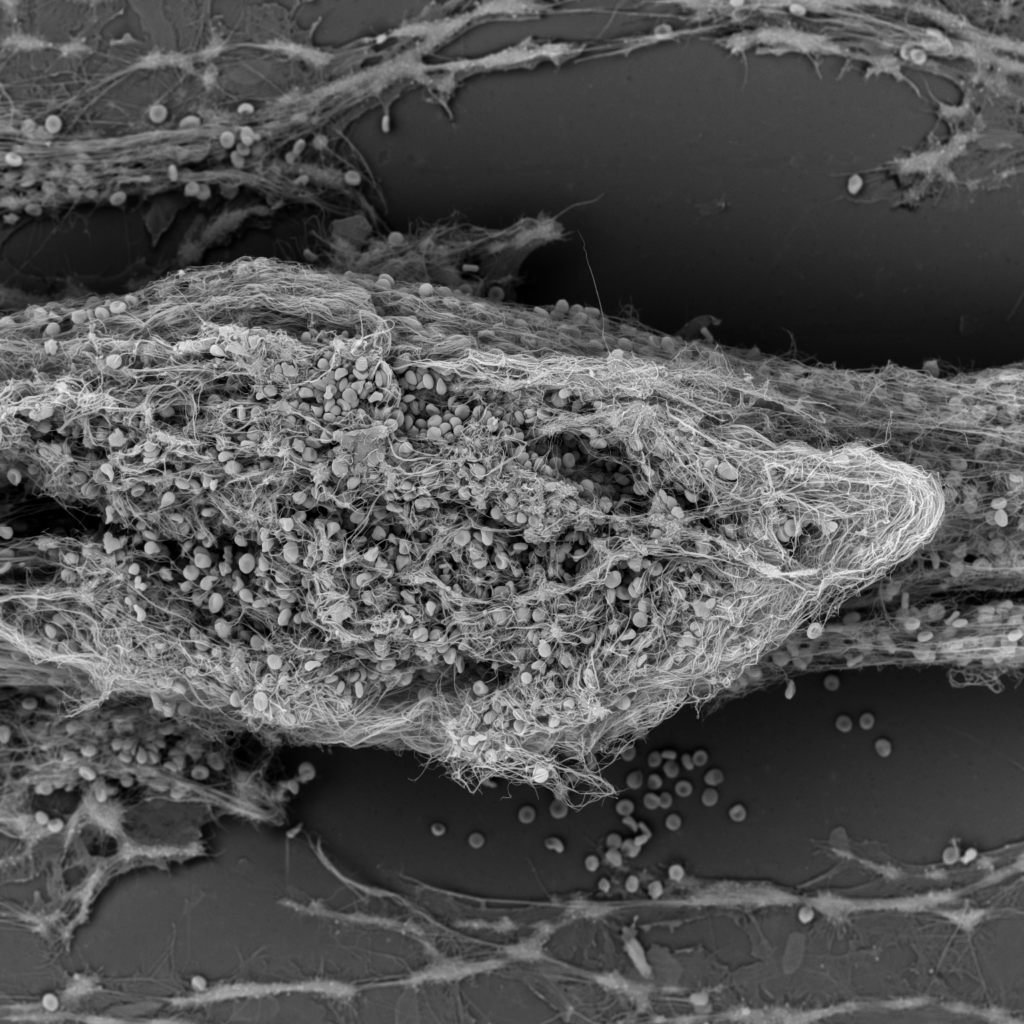
In a paper published in Clinical Pharmacology and Therapeutics, a Blood Vessel-Chip was developed to test a compound, a monoclonal antibody called Hu5c8, that had been shown to cause unwanted thrombosis in humans. Hu5c8 displayed promise as a treatment for lupus, but was ultimately terminated during clinical trials due to unexpected thrombotic and cardiovascular events it caused in patients. Unfortunately, these life-threatening side effects were not discovered during preclinical testing due to a lack of human-relevant models that could predict them. Using the Blood Vessel-Chip, researchers detected for the first time the pro-thrombotic effects of Hu5c8 using one clinically relevant dose of this compound in combination with human blood samples, and the data generated were consistent with previous clinical findings.
What’s perhaps even more interesting is that although previous studies have shown that thrombosis can be recreated in vitro, these results were limited to imaging data, since the formation of the clot made it impossible to obtain soluble samples. But in recent work, a team of researchers created a new chip design that enabled the ability to sample and analyze biomarkers from a clotted chip, allowing insight into mechanism of action of how Hu5c8 led to thrombosis.
Efficacy Testing of Anti-Platelet Drugs
With this chip design, researchers could induce thrombosis by stimulating endothelial inflammation, or by adding blood-borne, pro-coagulant factors. This approach created the ability to study the efficacy of anti-platelet drugs and is highlighted in the paper published in collaboration with Janssen Research & Development, the Wyss Institute at Harvard, and the University of Twente. This research shows how Organs-on-Chips technology enables the use of human-relevant models of drug-induced thrombosis, helps us dissect the molecular mechanisms that mediate this process, and it could also be used to study the efficacy of new anti-thrombotic therapeutics.