Featured session at Netherlands MPS Day, which took place on November 15, 2023.
Dr. Jeffrey Kroon from Amsterdam UMC discusses his team’s development of Organ-Chip models to better understand and treat cardiovascular diseases. Focusing on the role of endothelial cells in conditions such as atherosclerosis and ischemia-reperfusion injury after myocardial infarction, his group aims to identify novel therapeutic targets by recreating complex vascular environments in vitro. By manipulating endothelial metabolism, they hope to reduce inflammatory signaling and improve tissue outcomes.
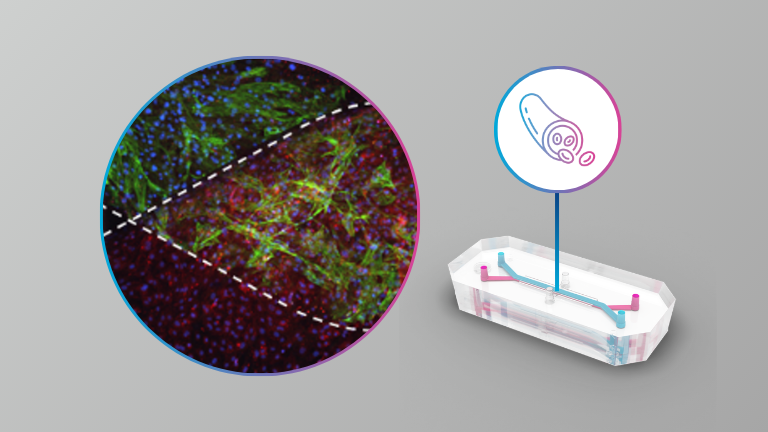
This presentation highlights three distinct microphysiological systems. First, a Plaque-on-a-Chip model integrates primary endothelial cells and macrophages separated by a porous membrane. When endothelial cells are inflamed—mimicking arterial plaques—secreted cytokines alter macrophage behavior, reflecting disease progression. This model enables detailed analysis of inflammatory cross-talk and the potential to dampen plaque development by metabolically modulating endothelial cells.
Second, a Heart-on-a-Chip model uses induced pluripotent stem cell (iPSC)-derived cardiomyocytes coupled to an endothelialized vascular channel. This setup simulates blood flow, nutrient delivery, and interactions between cardiomyocytes and endothelial cells. By introducing ischemia-reperfusion conditions, they can study how endothelial metabolism affects cardiomyocyte viability, beating frequency, and tissue damage. This system can inform strategies to mitigate reperfusion injury.
Lastly, Dr. Kroon showcases an Angiogenesis-on-a-Chip model, capturing the formation of new microvessels. Such a platform helps unravel how metabolic cues, growth factors, and inflammatory signals shape new vessel formation in atherosclerotic plaques or ischemic tissue.
Altogether, these human-relevant in vitro models provide a more nuanced understanding of the endothelial cell’s central role in cardiovascular disease. The combination of metabolic and inflammatory insights opens pathways toward targeted therapies that can stabilize plaques, reduce injury post-infarction, and improve vascular health.
Key learnings from this presentation include:
- Endothelial metabolism as a “volume control” for inflammation: Increasing endothelial glycolysis drives vascular inflammation in atherosclerosis, while reducing metabolic flux can calm inflammatory signals and prevent immune cell infiltration.
- Plaque-on-a-Chip for macrophage-endothelium crosstalk: Incorporating macrophages in a three-dimensional collagen matrix adjacent to inflamed endothelium offers a dynamic environment to test interventions that modulate plaque stability.
- Heart-on-a-Chip for ischemia-reperfusion injury: A model pairing iPSC-derived cardiomyocytes and endothelial cells simulates post-infarction conditions, enabling exploration of how endothelial factors influence cardiac tissue resilience, beating behavior, and stress responses.
- Angiogenesis-on-a-Chip for vascular remodeling: Controlled growth factor gradients and flow conditions help study new vessel formation, an important aspect of plaque progression and tissue repair.
- Toward translational impact: These advanced platforms bridge the gap between traditional cell culture and animal models, offering more predictive tools for developing therapies aimed at improving vascular health and cardiac outcomes.