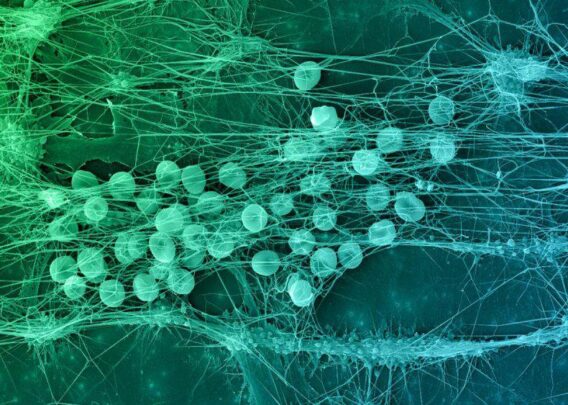
Data demonstrate differentiation of patient iPS cells into endothelial cells and neurons, using Organs-on-Chips to model the human brain’s microenvironment – providing a system to improve R&D for drug development for neurodegenerative diseases.
CAMBRIDGE, Mass. – Emulate, Inc., today announced that data on its Brain-on-Chip was presented by its collaborators at Cedars Sinai’s Board of Governors Regenerative Medicine Institute, who led the research and provided induced pluripotent stem (iPS) cells from patient samples to enable this new approach to model the human brain microenvironment. In the study, Emulate’s Brain-on-Chip demonstrated the physiologically-relevant modeling of the human brain’s microenvironment, including successful co-culture of human brain microvascular endothelial cells, neurons and astrocytes. The dynamic nature of the Brain-on-Chip facilitated the differentiation of iPS cells from patient samples into neurons, based on the creation of an appropriate microenvironment for growth and maturation of the brain cells. The findings from this collaborative research open up new applications for using Emulate’s Brain-on-Chip, in combination with patient-derived iPS cells from Cedars-Sinai, as a system to advance drug R&D in the field of neurodegenerative diseases and improve understanding of key mechanisms driving disease development. The data were presented at the Society for Neuroscience (SfN) 45th annual meeting taking place on October 17-21 in Chicago, Illinois.
The results achieved with the Brain-on-Chip using iPS cells define a novel system to assess the interactions between endothelial cells and neural tissue, forming the basis for new applications for disease-specific or patient-specific Organs-on-Chips using iPS cells derived from patients with neurodegenerative diseases, such as amyotrophic lateral sclerosis (ALS) or Parkinson’s disease.
“The data we have presented at the Neuroscience meeting represents just the beginning of the powerful ways that Cedars’ iPS cells and Emulate’s Organs-on-Chips can be combined to revolutionize drug development and health. Neurodegenerative diseases are one example of an area we can help identify and speed up the development of new solutions for patients,” said Clive Svendsen, PhD, senior author of the study and Director of Cedars-Sinai’s Board of Governors Regenerative Medicine Institute.
The new physiologically-relevant modeling of the human brain’s microenvironment with the new Brain-on-Chip system allows direct testing of how drugs cross the human blood brain barrier. This will allow pharmaceutical and biotechnology companies to test novel compounds’ ability to enter the brain in studies that precede clinical trials in human subjects. Using the Brain-on-Chip system may improve understanding of the mechanisms that regulate the blood-brain-barrier in order to enable novel strategies for the treatment of neurodegenerative disease. A major hurdle for neurological drug development is predicting how compounds will cross the blood brain barrier of humans. Animal models have limitations as there are significant species differences in the blood-brain-barrier that preclude extrapolation from animal data to humans.
With the demonstration of this new capability with a Brain-on-Chip system that models the human blood-brain-barrier, we look forward to working on drug development initiatives and to test how new medicines enter the brain and gain insights into disease mechanisms for new strategies to treat neurodegenerative disease.
— Geraldine A. Hamilton, PhD, President and Chief Scientific Officer of Emulate
“Emulate is actively advancing the applications of our Organs-on-Chips for neurodegenerative diseases, which is a therapeutic area where there are significant gaps in the drug development process that can be addressed with new human-relevant insights and predictive models, as exemplified by the data presented at the Neuroscience meeting,” said Geraldine A. Hamilton, PhD, President and Chief Scientific Officer of Emulate. “With the demonstration of this new capability with a Brain-on-Chip system that models the human blood-brain-barrier, we look forward to working on drug development initiatives and to test how new medicines enter the brain and gain insights into disease mechanisms for new strategies to treat neurodegenerative disease.”
Organs-on-Chips for Emulating Human Biology
Based on our Organs-on-Chips technology, which places living human cells in microengineered environments, Emulate has developed an integrated system that provides a window into the inner-workings of the human body. Our living products set a new standard for predicting human response, with greater precision and detail than today’s cell culture or animal-based testing. Emulate’s Organs-on-Chips contain tiny hollow channels lined by living human cells and tissues cultured under continuous fluid flow and mechanical forces, such as cyclic breathing and peristalsis, which recreate the microenvironment experienced by cells within the human body. Each Organ-on-Chip can contain tens of thousands of cells and is approximately the size of a USB memory stick. Organs-on-Chips are miniaturized living systems that represent the smallest functional unit of an organ that effectively recapitulate organ-level physiology and disease responses. Multiple Organs-on-Chips, such as lung, liver, intestine, kidney, skin, eye, and blood-brain-barrier, can be linked together by flowing human blood or nutrient-containing liquid to create a “Human-Body-on-Chips” that closely replicates whole body-level responses.
About Emulate
Emulate, Inc. is a private company that creates living products for understanding how diseases, medicines, chemicals, and foods affect human health. By setting a new standard for recreating true-to-life human biology, Emulate is advancing product innovation, design and safety across a range of applications in drug development, personalized health, agriculture, cosmetics and chemical-based consumer products. Emulate continues to develop a wide range of Organs-on-Chips, disease models and diagnostics through collaborations with industry partners and internal programs. By combining our Organs-on-Chips system with individuals’ stem cells, Emulate is accelerating progress toward a new era of precision medicine and personalized health. For more information, visit www.emulatebio.com.
Media Contact:
Kathryn Morris
The Yates Network
Tel: 845-635-9828
kathryn@theyatesnetwork.com