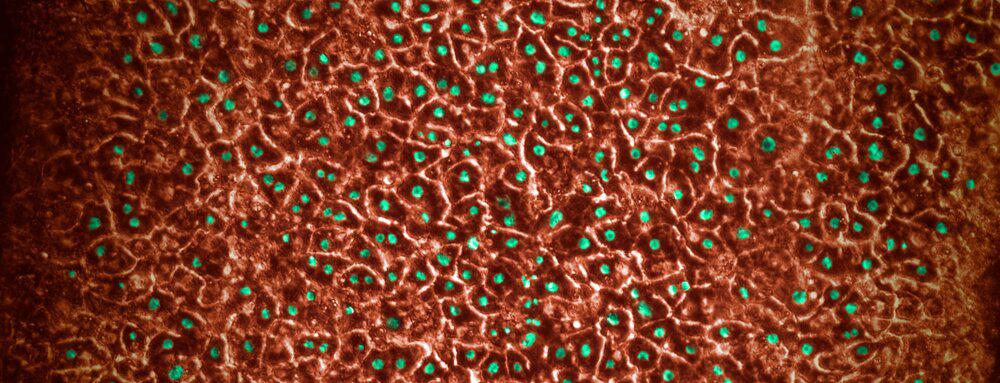
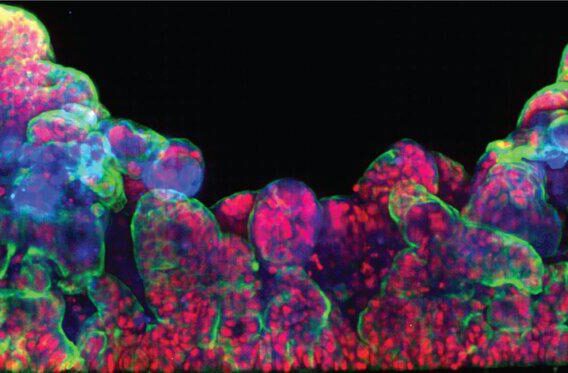
Before a drug comes to market, it is subjected to rigorous tests to ensure its safety and efficacy. However, many standard safety tests for drug candidates involve rodent and non-rodent animal models which cannot always accurately predict which drugs might cause drug-induced liver injury (DILI) in humans. To increase predictive confidence ahead of clinical studies, a species-specific, multicellular, in vitro hepatotoxicity assay was developed, using our Liver-Chip, to characterize human toxicities, including responses that may have not been accurately predicted by animal models.
SNAPSHOT
Research Area: Hepatotoxicity testing
Organisms: Human, dog, rat
Sample Types: Human, dog, rat Hepatocytes, liver sinusoidal endothelial cells, Kupffer cells, and hepatic stellate cells
Research Question: Can a multispecies Liver-Chip predict liver toxicity and inform human relevance of liver toxicities detected in animal studies?
EXPERIMENTAL OVERVIEW
Emulate Organs-on-Chips technology was used to create rat, dog, and human Liver-Chips. Primary rat, dog, or human hepatocytes were seeded in the upper parenchymal channel within an extracellular matrix (ECM) layer on top of an ECM-coated, porous membrane that separates the two parallel microchannels. Species-specific rat, dog, or human liver sinusoidal endothelial cells (LSECs), with or without Kupffer cells and/or stellate cells, were cultured on the opposite side in the lower vascular channel. These quadruple-cell chips were characterized by albumin secretion and Cytochrome P450 enzymatic activity prior to drug toxicity testing.

RESULTS
Studying species-specific drug toxicities
To explore whether the rat, dog or human liver chips could be used to predict species-specific DILI responses, the study evaluated hepatotoxic effects induced by bosentan, a dual endothelin receptor antagonist. Due to species differences in the bile salt export pump (BSEP) efflux transporter, bile salts accumulate within hepatocytes upon exposure to bosentan, causing cholestasis in humans, but not in rats or dogs. The results showed that the mechanisms of drug induced liver injury can be dissected using the Liver-Chip.
Detection of diverse phenotypes of hepatotoxicity
The species-specific, quadruple cell Liver-Chips were tested with tool compounds that are known to cause different kinds of toxicity and provided insights into the drug mechanism of action. In brief, the experiments were designed to detect Kupffer cell depletion, and markers of steatosis and fibrosis, with the results suggesting that the Liver-Chip could be a useful model for predicting toxicities. In addition, candidate drugs that show hepatotoxicity in dog and rat models can be screened for potential risk assessment in the human quadruple cell Liver-Chip.
Towards a more accurate model for pre-clinical screening
One of the most difficult forms of hepatotoxicity to predict relates to DILI responses that are often missed during preclinical and early clinical testing. The human Liver-Chip was exposed to TAK-875, a G protein coupled receptor 40 agonist that was discontinued in phase 3 trials due to DILI in a few individuals. The experiments showed that continuous and prolonged exposure caused mitochondrial dysfunction, oxidative stress, formation of lipid droplets, and an innate immune response, all of which are harbingers of DILI for susceptible patients. These results highlight the advantage of the Liver-Chip for assessing the pathophysiological consequence of reactive metabolite formation, which has been strongly associated with DILI.
CONCLUSION
This study explores whether species-specific, in vitro hepatotoxicity assays using Liver-Chips can provide a greater understanding of the mechanisms of hepatotoxicity in animal models, which may lead to reduction in the number of animal studies to provide accurate risk assessment for drug candidates. The Liver-Chip detected diverse types of liver toxicity, including hepatocellular injury, steatosis, cholestasis, and fibrosis. The study has shown that species-specific Liver-Chips have potential future applications for hepatotoxicity testing, disease modeling, biomarker identification. This experimental approach could also be used to study whether toxicities observed in animal models are translatable to humans, and to understand human cellular variability with regards to drug response.
REFERENCE
Jang, KJ et al. Reproducing human and cross-species drug toxicities using a Liver-Chip. Sci Transl Med 019 Nov 6;11(517).